News Releases
News Release
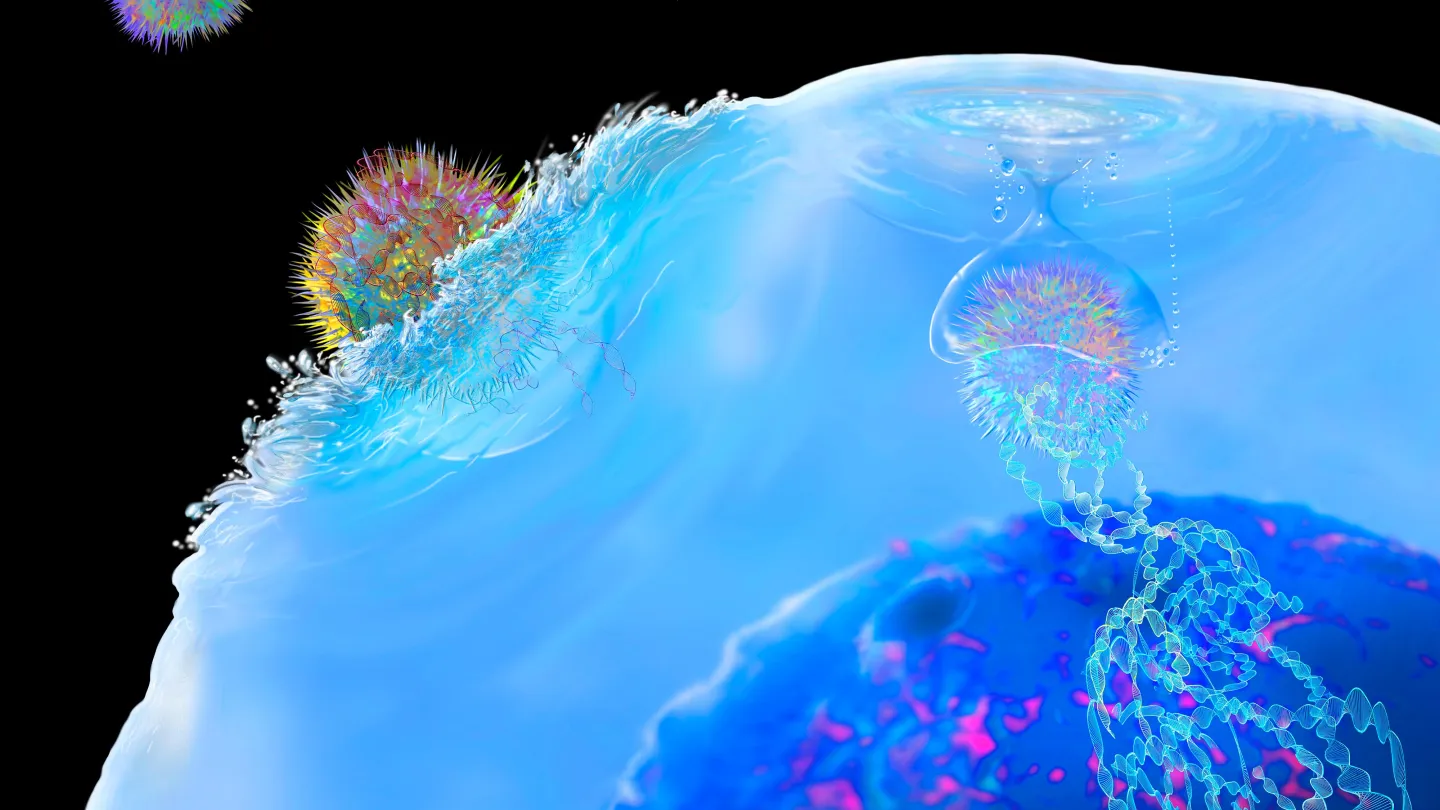
New Study Identifies Signature in Blood to Better Predict Type 2 Diabetes Risk
January 14, 2026
News Release
Dr. Martha E. Pollack to Deliver Keynote Address at Einstein’s 2026 Commencement
January 9, 2026
News Release
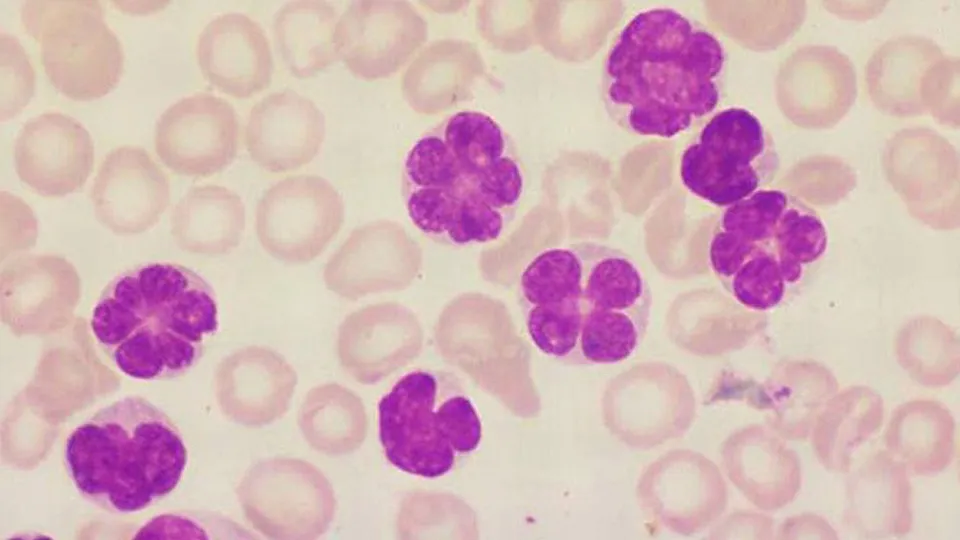
Night Waking Impacts Cognitive Performance Regardless of Sleep Duration
December 18, 2025
In the News
Header
Experts for Media
Header
Experts for Media
Research
Education & Training